1. The challenge
Rehabilitation is more than physiotherapy.
In early stroke rehabilitation, clinicians often observe declining motivation over time. At the same time, progress frequently lacks transparency: assessments rely on subjective impressions rather than objective longitudinal data, and complications may only be detected at a late stage.
Patient engagement is therefore not a “soft” factor. It is a potentially modifiable driver of clinical outcomes.
Yet in routine clinical practice, scalable tools are still lacking to systematically measure motivation, integrate digital biomarkers, and actively involve patients in their rehabilitation process.
2. The study approach
Sensor-S: A multicenter randomized controlled trial
The Sensor-S study (“Effect of Wearable Sensors on Patient Engagement and Motivation in Post-Stroke Rehabilitation”) is a multicenter randomized controlled trial (RCT) involving:
- 156 patients
- two study centers (Charité Berlin and Brandenburgklinik)
- two intervention phases (inpatient and outpatient)
- six months of follow-up
The study investigates whether patient motivation can be objectively captured using digital sensor technologies, and how this influences the evaluation of rehabilitation trajectories.
3. The technological setup
Multimodal data collection via the D4L Collect study app
The intervention combines multiple digital data sources through the D4L Collect study application.
Smartwatch (Samsung Galaxy Watch 6)
→ Heart rate, activity, sleep, step count
CGM (Dexcom G7)
→ Continuous glucose monitoring every 5 minutes
IMUs (Inertial Measurement Units)
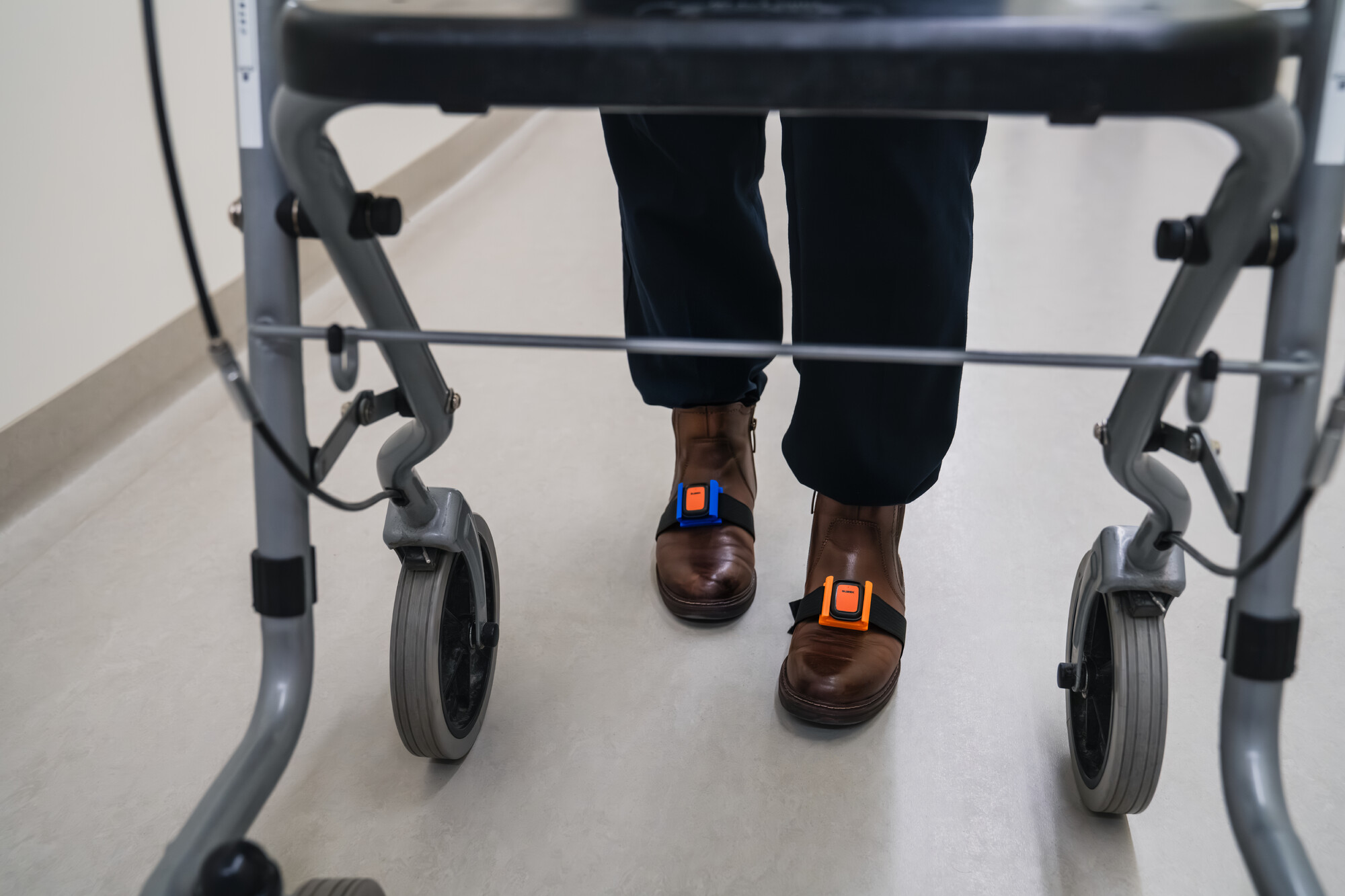
→ Gait, Timed Up & Go, Sit-to-Stand, Spasticity measurement
Mobile D4L Collect App (Android, z. B. Google Pixel 6)
→ Daily reflections
→ Motivation assessment
→ Sleep & pain surveys
→ Daily & weekly reports
→ Motivational prompts
Patient-centered intervention
The app integrates SMART-based goal setting with daily reflections (text-based entries), motivational prompts, and structured reports including metrics such as step count, activity level, and heart rate (retrospective).
These reports are visualized in a patient-friendly format, an essential component for strengthening patients’ sense of self-efficacy.
Accompanying visual documentation, such as sensor placement, gait analysis procedures, and interactions with the study app, helps illustrate these processes and improves accessibility for a professional audience.

4. The infrastructure behind it
Why infrastructure matters more than individual sensors
Sensors embedded in smartphones and wearable devices generate data.
Clinical studies, however, require structured, interoperable, and analyzable datasets. The Sensor-S study uses the native mobile application D4L Collect, developed by data4life, to collect and integrate data from multiple sources.
Key architectural features include:
1. Separate pseudonyms
Study ID (DTS Charité)
Separate app ID
No PII in the app account
2. OAuth2 integration with the Dexcom EU API
Secure CGM data transmission
Retrospective display in the D4L Collect interface
3. Encryption
at rest
in transit
server location Germany
4. Structured data collection
PROMs (PAM-13, PAREMO, EQ-5D-5L, etc.)
IMU raw data
CGM raw data
Smartwatch data
5. Compliance tracking
Monitoring of the daily questionnaire rate
Evaluation of app interaction patterns
5. Implications for clinical research
Die Sensor-S-Studie zeigt, dass Sensoren in Smartphones und Wearables im klinischen Kontext mehr sind als passive Datensammler. Sie fungieren zugleich als Feedback-Instrumente, Motivationsverstärker und potenzielle Quelle digitaler Biomarker. Damit werden sie Teil des Interventionsdesigns – nicht nur technische Begleiter der Datenerhebung.
The Sensor-S study shows that sensors in smartphones and wearables serve a broader role in clinical settings than merely collecting passive data. They can also function as feedback mechanisms, motivational drivers, and potential sources of digital biomarkers. In this way, they become an integral part of the intervention design, not just technical companions to data collection.
However, the crucial factor is the interaction between intervention and infrastructure. A single device does not change behavior. Effective digital interventions emerge only when structured reporting, clear goal definition, daily app interaction, and systematic compliance monitoring are combined. Sensors generate data and infrastructure makes that data usable.
Digital biomarkers are derived from carefully structured datasets. In the context of CGM data, this includes parameters such as glycemic variability, CVBG, time in range, or mean amplitude of glycemic excursion. For inertial measurement units (IMUs), relevant metrics include gait parameters, stride length, angular velocity, or spasticity quantification.
These data can only be analyzed if timestamps are harmonized, identities are managed separately, raw data remain accessible, and export formats support statistical analysis.

Finally, scalability begins at the level of study design. With both inpatient and outpatient intervention phases, including remote setup and remote visits, the Sensor-S study demonstrates that digital clinical studies can extend beyond the hospital environment. Technically, organizationally, and conceptually.
Planning a digital study?
Talk to us.
D4L Collect enables structured, secure, and scalable data collection for clinical studies—from pilot projects to multicenter RCTs.
Simply send an email to b2b@data4life.care.
The information in this article reflects the current state of scientific knowledge at the time of publication and has been prepared to the best of our knowledge and belief. However, this article cannot replace professional medical advice or diagnosis. If you have medical questions, please consult your general practitioner.
The contents of this article reflect the current scientific status at the time of publication and were written to the best of our knowledge. Nevertheless, the article does not replace medical advice and diagnosis. If you have any questions, consult your general practitioner.